Study population
Between 2 March 2021 and 13 June 2022, we completed a prospective, longitudinal, observational household cohort study of SARS-CoV-2 incidence and immunity in The Gambia (Transmission of Respiratory Viruses in Household in The Gambia: TransVir; clinicaltrials.gov NCT05952336). The Gambia is a small country in West Africa, ranked 174th by the United Nations Human Development Index in 2021. The climate is sub-tropical, with a short rainy season from June to October each year. The study was conducted at two urban sites, the West Coast Region and Kanifing Municipality of The Gambia. Both sites had households that had previously participated in studies conducted at the Medical Research Council Unit The Gambia at The London School of Hygiene & Tropical Medicine (MRCG). These sites were selected due to established relationships with these communities that facilitated participant recruitment during the COVID-19 pandemic.
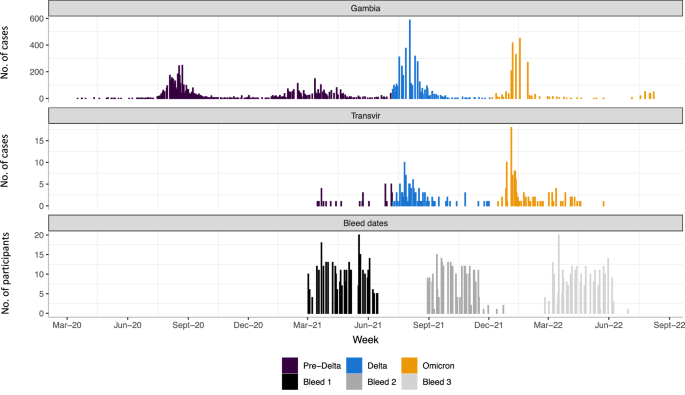
349 participants were recruited from 52 households, a median of 6 individuals per household (IQR 5–8); Table 1. Forty-one participants were children under 5 years old (11.7%), 153 were 5–17 years old (43.8%), 130 were 18–49 years old (37.2%) and 25 were ≥50 years old (7.2%); with 201 (57.6%) female participants. Recruitment commenced during the 2nd SARS-CoV-2 wave in The Gambia (Fig. 1), with baseline (V1) visits completed prior to the Delta (3rd) wave and most 6-month (V2) visits completed prior to the Omicron BA.1 (4th) wave10.
Top panel shows case numbers reported in The Gambia (WHO COVID-19 Dashboard. Geneva: World Health Organization, 2020. Available online: https://covid19.who.int/). Middle panel shows RT-PCR-positive events in the study cohort. Cases are coloured by SARS-CoV-2 variant period (pre-Delta, Delta, Omicron), based on dates derived from wider sequence data from The Gambia, corresponding to weeks when >50% of sequenced isolates were first Delta (7 July 2021) or Omicron-lineage (4 December 2021) viruses. Bottom panel shows a number of individuals providing samples for serological analysis during V1 (bleed 1; baseline), V2 (bleed 2; 6-month visit) and V3 (bleed 3; 12-month visit) timepoints. RT-PCR reverse-transcriptase polymerase chain reaction.
Binding antibody responses to SARS-CoV-2 Spike (S) and Nucleocapsid (N) targets were measured at V1, V2 and V3 (12-month visit). SARS-CoV-2 spike antibody seropositivity increased from 56% at V1 to 84% at V2 and 94% at V3 (Table 1). A similar pattern was observed for antibodies to nucleocapsid (47% at V1, 69% at V2, 88% at V3). No participants had received SARS-CoV-2 vaccines at study entry. By 6 months of follow-up, 32 participants (6.9%) had been vaccinated, increasing to 48 participants at 12 months (14%). A median of 45 scheduled visits was completed per participant (IQR 39–48), with 338 (96%) completing more than 5 (10%) scheduled visits and included in further analyses.
Incidence and risk factors for SARS-CoV-2 infection
The prespecified primary objectives were to establish the incidence of SARS-CoV-2 infection during the study period and to determine differences in risk between children and adults. Secondary objectives were to determine the secondary attack rate of SARS-CoV-2 in households, the sero-incidence and cumulative seroprevalence of SARS-CoV-2, the frequency of asymptomatic SARS-CoV-2 infection, and risk factors associated with SARS-CoV-2 infection.
Combined throat and nose flocked swabs (TNS) were collected for 52 weeks regardless of symptom status for SARS-CoV-2 reverse-transcriptase polymerase chain reaction (RT-PCR). Of 14569 visits with an RT-PCR result, 334 were positive (Fig. 2). A SARS-CoV-2 RT-PCR-confirmed episode was defined as ≥1 SARS-CoV-2 RT-PCR-positive TNS, with no previous RT-PCR-positive sample in the prior 28 days. Increases in antibody titre between sequential bleeds (V1–V2, V2–V3) in the absence of a positive RT-PCR result were used to define infection episodes missed by RT-PCR, with only nucleocapsid antibodies assessed for participants vaccinated between bleeds. One hundred and eleven PCR-negative but serologically-defined infection episodes were identified, equalling a total of 381 infection episodes (270 PCR-positive; 111 PCR-negative) and an incidence rate of 1.34 infections-per-person-year (95% CI 1.21–1.48).
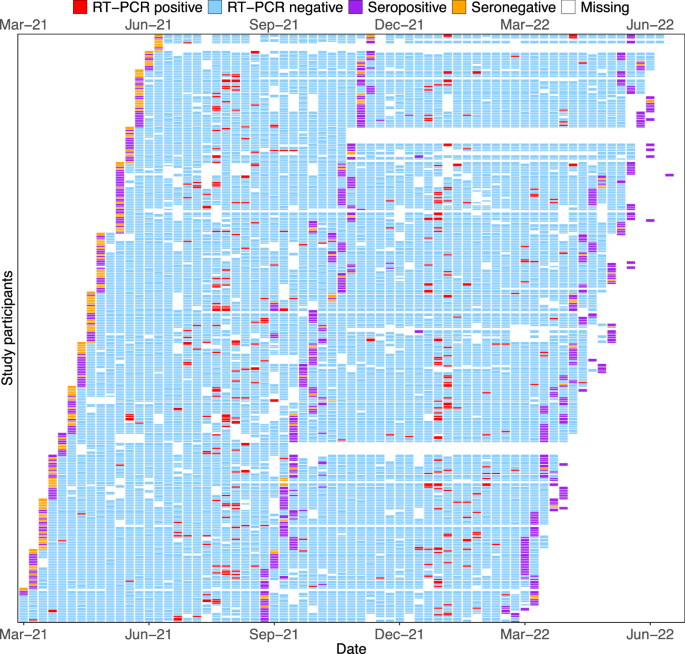
Each row represents a participant numbered consecutively, grouped together in households. Columns represent follow-up visits. A participant’s follow-up time was divided into weekly breaks. Given the asynchronicity of recruitment, visits are not necessarily aligned with the same weekly breaks for all participants. As a result, a small number of weekly breaks for some participants may contain two visits. In such a situation, the result of the visit is determined hierarchically (seropositive, seronegative, RT-PCR-positive, RT-PCR-negative, missing). Seropositive and seronegative relate to positive and negative results in a serum anti-spike antibody assay. RT-PCR reverse-transcriptase polymerase chain reaction.
There were 270 first infections and 111 re-infections (99 second and 12 third infections) during study follow-up. Of the 270 RT-PCR-positive infections, 236 were RT-PCR-positive during one visit only, 18 were positive for 7-14 days, 12 were positive for 14–21 days, and 4 episodes were positive for 21 days (maximum 28 days). SARS-CoV-2 infections were stratified into ‘pre-Delta’, ‘Delta’ and ‘Omicron’ calendar periods based on weeks when >50% of sequenced isolates in The Gambia were first Delta (7 July 2021) or Omicron-lineage (4 December 2021) viruses10. Attack rates were 11.8% (95% CI: 8.6–15.7) during the Pre-Delta period included in the study (prior to 7 July 2021), 44.6% (95% CI: 39.3–50.3) during the Delta period (7 July 2021 to 4 December 2021) and 56.7% (95% CI: 51.0–62.3) during the Omicron period (after 4 December 2021). In addition, Omicron had a higher attack rate amongst previously infected participants (56.3%, 95% CI: 50.3– 62.2) compared to Delta (26.7%, 95% CI: 21.6–32.3). Seven RT-PCR-positive episodes at baseline were excluded from incidence-based analyses as the time of infection was not known.
After adjusting for prior infection status, variant period, vaccination status, and household size, children under 5 years old had a lower hazard of infection than 18–49-year-olds (aHR 0.48, 95% CI: 0.31–0.74, p = 0.0002, Table 2). Children <5 years old had a lower hazard of infection compared to 18-49-year-olds in both Delta- and Omicron periods. These results were consistent in all sensitivity analyses using only RT-PCR-positive infection episodes, different serological definitions for RT-PCR-negative infections, and considering all RT-PCR-positive events within 90 days of a first positive result part of the same infection episode (Tables S2–S4).
V1 spike antibody status was used to assign the number of prior infections at the start of the study, with the presence of spike antibody at V1 assumed to be from one prior infection. During the study follow-up, prior infection status increased following an RT-PCR-confirmed infection or the estimated date of an RT-PCR-negative, serologically defined infection. In univariate and multivariate analyses, there was strong evidence that prior SARS-CoV-2 infection was associated with a reduced hazard of subsequent infection (p < 0.0001, Table 2, Fig. 3A). Compared to those previously uninfected, 1 prior infection was associated with a 58% lower hazard of infection (adjusted hazard ratio (aHR) 0.42, 95% confidence interval, CI, 0.32–0.56), with ≥2 prior infections associated with an 87% reduction in infection hazard (aHR 0.13, 95% CI: 0.09–0.20, p < 0.0001).
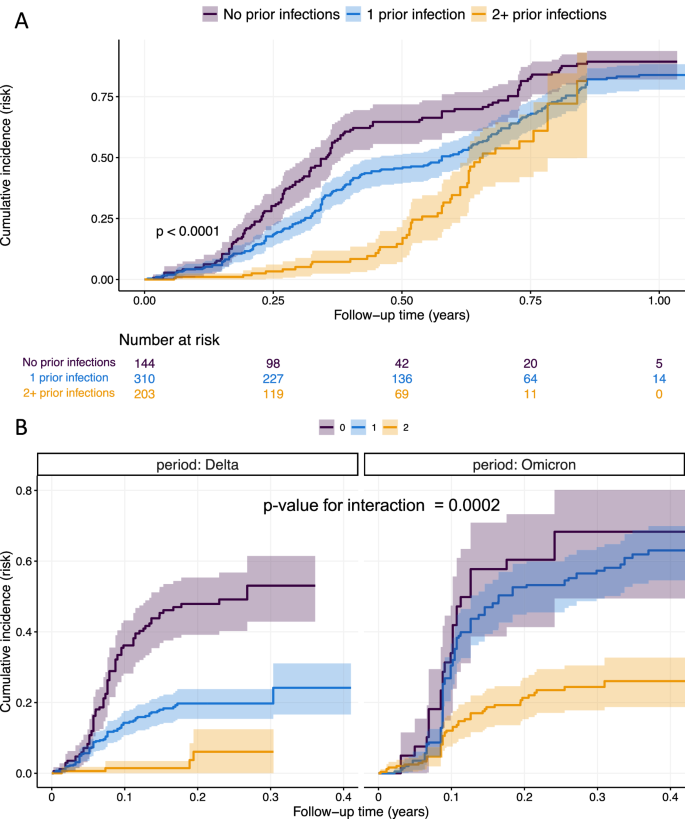
The cumulative incidence over follow-up time is expressed as a risk (number infected/number at-risk) and displayed for A the whole study period or B stratified by Delta- and Omicron-variant periods. P-value calculated by log-rank test for association between prior infection status and infection incidence (A) and for interaction between Delta and Omicron periods (B). The test for interaction assesses whether the association between prior infection status and infection incidence is modified by the circulating variant (i.e., is the effect of one prior infection on infection incidence different between Delta and Omicron). All p-values are two-sided. Shaded area represents the 95% confidence interval.
The association between prior infection status and infection hazard was modified by the dominant variant (p-value for interaction = 0.0002). Compared to previously uninfected individuals, those with one prior infection had a lower hazard of infection in the Delta period (aHR 0.28, 95% CI: 0.18–0.42) but not in the Omicron period (aHR 0.74, 95% CI: 0.49–1.13). In contrast, those with ≥2 prior infections had a lower hazard of infection in both Delta- and Omicron-variant periods (Table 2, Fig. 3B). These findings were consistent in all sensitivity analyses (Tables S2–S4).
To assess whether the increased infection hazard during Omicron was due to a longer interval between infections (and therefore increased waning of immunity) an additional analysis was conducted. The time elapsed between consecutive infection episodes was calculated, with a date of first infection imputed by probability sampling for those sero-positive at baseline. Infection status was categorised as: “No prior infections”, “1 prior infection within 90 days”, “1 prior infection 90 or more days ago”, “2 or more prior infections, with the most recent within 90 days”, and “2 or more prior infections, with the most recent 90 or more days ago”. Compared to participants with no prior infections, participants with 1 prior infection in the last 90 days had a decreased infection hazard during the Delta (aHR 0.29, 95% CI 0.12–0.67) but not during the Omicron wave (aHR 0.73, 95% CI 0.28–1.91, Table S1). Similarly, for participants with 1 prior infection that occurred more than 90 days ago, there was a decreased infection hazard during the Delta (aHR 0.28, 95% CI 0.19–0.43) but not during the Omicron wave (aHR 0.75, 95% CI 0.50–1.14, Table S1). There remained strong evidence of interaction between the incidence of infection, prior infection status, and the dominant variant (p < 0.0001).
Symptom status of RT-PCR-positive infections
At each visit, the presence of influenza-like symptoms in the preceding 7 days was assessed. A symptomatic SARS-CoV-2 episode was defined as an RT-PCR-positive episode associated with the presence of ≥1 symptom consistent with COVID-19 from 1 week before to 1 week after the first RT-PCR-positive visit. Of 263 RT-PCR-positive infections during follow-up, only 41 (15.6%) were associated with symptoms (6 missing symptom data). Cough was the most frequently reported symptom (25/41), with headache (16/41) and fever (15/41) also common (Table S5). Prior infection status, age, and variant period were not associated with the presence of symptoms (Tables S6 and S7).
Household cumulative infection risk (HCIR)
A SARS-CoV-2-positive household cluster included all individuals testing SARS-CoV-2 RT-PCR-positive in a household within ≤14 days of any RT-PCR-positive episode. Households experienced a median of 2 clusters during the study period (IQR: 1 – 3, maximum 5), with 51/52 households having at least 1 cluster. One hundred and eighteen household clusters were identified during the study, with 93 resulting from a single index case (the first RT-PCR-positive participant in a cluster) and 25 with multiple index cases (two or more participants testing RT-PCR-positive on the first day of a cluster). Of the 93 single index cases initiating clusters, 14 (16%) were symptomatic (5 missing symptom data), 3 were under 5 years old, 46 were 5–17 years old, 38 were 18–49 years old and 6 were >50 years old. 466 participants were exposed in the 93 single-index clusters, with 43 subsequently testing SARS-CoV-2 RT-PCR-positive (an HCIR of 9.2%). Four hundred and forty-four participants underwent RT-PCR testing within one serial interval (14 days) of the index case positive RT-PCR, of which 35 tested positive (a secondary attack rate, SAR, of 7.9%). There was no evidence of any association with characteristics of the index case, variant period, or household with an increased odds of transmission to exposed participants (Tables S8 and S9). There was weak evidence in the univariate HCIR analysis that positive serology in the index case was associated with lower odds of onward transmission compared to indexes with negative serology (OR 0.47, 95% CI 0.21– 0.99, p = 0.05, Table S8). This finding was not replicated in the univariate SAR analysis (OR 0.57, 95% CI 0.24–1.25, p = 0.16, Table S10). After adjusting for the SARS-CoV-2 serology status of the exposed household contacts, no association remained in either HCIR (aOR, 0.57, 95% CI 0.22–1.14, p = 0.06) or SAR analyses (aOR 0.57, 95% CI 0.23–1.30, p = 0.23, Tables S9 and S11).
Credit: Source link